If you’ve heard that ion exchange can improve the quality of treated water, you might be wondering how it works. Whether you utilize water from a private well or the municipal system, it’s essential to take all necessary measures to ensure the water is safe and clean.
To help you decide whether or not an ion exchange water filter is right for your home or business, we’ve compiled all the information you need to make an informed decision.
What Exactly Is Ion Exchange?
Ion exchange is a water treatment technology that is often used for water softening or demineralization inside an ion exchange resin [1]. Notably, it can help soften hard water by removing magnesium and calcium ions.
It’s also utilized in water purification operations, including deionization, dealkalization, denitrification, and disinfection.

What Is Ion Exchange Resin
When used for water softening, ion exchange resins swap out the water’s cations for sodium ions of sodium chloride. It also replaces water’s anions for chloride ions. Furthermore, they can be used to demineralize water by exchanging the cations for H+ ions and the anions for OH− ions.
Which Ions Are Exchanged?
There are two types of ions, which we call anions and cations.
Anion Exchange
To execute the anion exchange, a positive charge must be applied to the solid material in order to attract negatively-charged cations. Then, the solid resin attracts the unwanted cations and releases the desirable cations into the water during the ion exchange process.
Cation Exchange
In cation exchange, the solid substance is negatively charged, and the favorable and unfavorable cations are positively charged.
The process of cation exchange is often utilized in water softeners, with sodium chloride or hydrochloric acid serving as the desirable cations and hardness minerals serving as the unwanted cations.
The Ion Exchange Regeneration Cycle
There’s a limit to how many cations or anions an ion exchange resin can contain. At first, the resin is loaded with desirable cations, but after a period of time, these cations are leached out into the water and replaced with undesirable ions.
The resin becomes overloaded with unwanted ions and deprived of desirable ions. Thus, this calls for regeneration. This is where undesirable ions are flushed out of the resin, and the desired ones are added.
Water is typically used to wash out the resin during the regeneration process. Then the resin is saturated with the desirable ions, and they attach to the surface. This procedure is repeated until the resin is again filled with desirable ions.
Related Article: Best Water Softener Systems
How Is Ion Exchange Used For Water Treatment?
The ion exchange technique is a popular method of treating drinking water. In order to purify water, specific resins are used to eliminate a variety of common contaminants. Nitrates, arsenic, calcium, magnesium, borons, perchlorates, and uranium are some of the contaminants that are eliminated in this process.
There are two types of water treatment technologies that use ion exchange to improve water quality: water softeners and water filters.
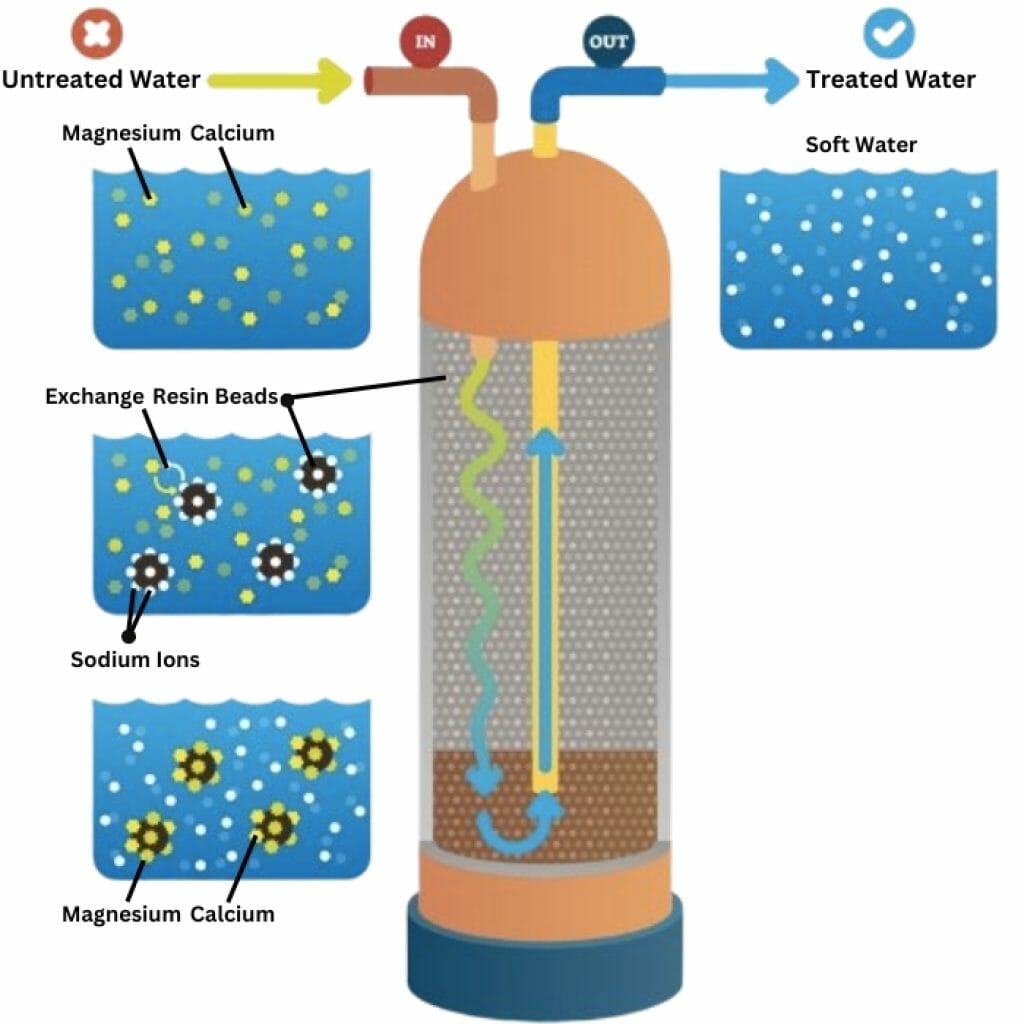
Ion Exchange Water Softener
Through the process of ion exchange, the positively charged calcium and magnesium ions in water are exchanged with the positively charged sodium. This is commonly done in traditional salt-based water softeners. This process occurs in a bed of negatively charged resin.
A water-softening ion exchange system consists of a sodium tank and a resin tank. In a salt brine solution, sodium ions are pulled into the resin tank because sodium is drawn to the opposite charge of the resin beads.
When water flows through the resin tank, an exchange softening process takes place, and the sodium in the beads stays in the resin bed. During the water-softening process, the calcium and magnesium ions are removed to produce salt-softened water.
When the resin beads are entirely loaded with magnesium and calcium ions, a regeneration cycle is run to remove the minerals and replace them with new sodium ions.
Ion Exchange Water Filter
Ion exchange filter media is like ion resin in a water softener but on a smaller scale. Similarly, in the exchange reaction, undesirable ions in drinking water are replaced by desired ions.
Cation exchange resins are used by a number of different manufacturers in their filters, including Brita in its pitcher filters. The positive charge of calcium, magnesium, lead, and copper ions are exchanged for the positive charge of hydrogen ions in a cation exchange resin.
The purpose of an ion exchanger is to convert positively charged ions (such as sulfate, phosphate, and nitrate) into negatively-charged hydroxide or chloride ions.
How Effective Is Ion Exchange Water Treatment?
The Environmental Protection Agency (EPA) has recognized ion exchange as the “Best Available Technology” (BAT) and the Small System Compliance Technology (SSCT) for dealing with
- Radium
- Uranium
- gross alpha
- Beta
- Photon emitters.
Anion exchange is used to get rid of
- Alkalinity
- Nitrates
- Arsenic
- Chromium-6
- Cyanide
- Perchlorate
- Sulfate
- Uranium
And a cation exchange is used to get rid of
- Calcium
- Magnesium
- Iron
- Manganese
- Barium
- Radium
- Strontium
Pros and Cons Of Ion Exchange
Ion exchange water filters are reliable and effective, making them a good choice for residential and office water purification. Let’s look into the pros and cons.
Pros
- An effective technique for softening water. To treat hard water, nothing beats the tried-and-true method of ion exchange water softening.
- Has a high rate of success. When it comes to purifying liquids from unwanted charged ions, ion exchange is remarkable. Once the correct water parameters are established, the treatment is assured to be effective.
- It can eliminate most contaminants. Various ion exchange resins are available to remove hardness minerals and other contaminants that could potentially be found in drinking water.
- Less costly to maintain. If you use an ion exchange system in your home, you probably won’t spend more than one hundred dollars a year on maintenance, as most of them only need replenishment with desirable ions every few weeks.
- Has a long lifespan. Most ion resins have a service life of 8-12 years before they need to be replaced.
Cons
- Water quality issues may have an impact. Ion exchange resins can scale, clog, or get contaminated by specific water contaminants.
- Makes water more acidic. The exchange process often eliminates alkaline minerals, lowering water’s pH and raising the possibility of metal leaching.
- Needs regeneration. If the resin is replaced often the exchange process will work. Regeneration requires regular availability of favorable ions.
- There is still some maintenance required. Although ion exchange takes less maintenance than specific water treatment systems, it still requires topping up desirable ions or changing a filter.
- Produces water wastage. A salt-based IE water softener wastes liters of water during regeneration. Sadly, this must be done.
FAQs For Ion Exchange Process
Is it safe to drink ion exchange water?
Drinking water that has been through an ion exchange process is generally safe for consumption. After all, it is one of the best water treatment methods to eliminate contaminants.
What is the best ion exchange resin to use?
To remove iron and calcium ions, choose a cation exchange system. If you want to get rid of carbonates, chlorides, and nitrates, use anion exchange.
Are ion exchangers expensive?
Off-site services might cost $40 to $100 per cubic foot, depending on flow rates and resin changes.
Is ion exchange better for filtration or softening?
It all depends on what you intend to get rid of. An ion exchange system can effectively remove certain ionic pollutants and impurities.
What’s the difference between ion and anion exchange?
Cation resins are positively charged, while anion resins are negatively charged (anion). Because of this, they can be used to effectively eliminate certain types of contaminants.
Article Sources
Freshnss uses only the highest-quality sources to support the facts used in our articles including: government organizations, independent studies, peer-reviewed journals, and lab testing results. Read our editorial review guidelines here to learn more about how we verify and fact-check our writing to keep our content reliable, accurate, and trustworthy.
- Drinking Water Treatment Water Softening (Ion Exchange), University of Nebraska–Lincoln Extension




